Fluorescence Recovery After Photobleaching (FRAP) is nowadays considered as the mode widely used method to monitor protein and organelle dynamics. This application note introduces the use of the Inscoper scanFRAP solution to explore the cinetic of endoplasmic reticulum in live cells.
FRAP experiment on HeLa cells.
Biological context
The endoplasmic reticulum (ER) is a membrane-bound organelle found throughout the cytoplasm of all eukaryotic cells. The ER plays a central role in many biological processes, including protein synthesis, glucose-lipid metabolism, signaling, and calcium dynamics (Öztürk et al, 2020). This abundant structure is formed and maintained by a permanent remodeling process that involves the synthesis, transport, and fusion with pre-existing ER tubules. Dysfunction of the ER could induce severe diseases such as neurodegenerative disease, cancer, or metabolic diseases (Ozcan & Tabas, 2012; Roussel et al, 2013; Oakes, 2020). Thus, the live imaging of the ER dynamics became a gold standard technique in order to better understand the pathophysiological pathways involved.
FRAP technique for live cell imaging
In live cell imaging, biologists commonly use fluorescent markers to track organelles and/or proteins. However, these fluorophores can permanently lose their ability to emit fluorescence when the excitation light is very intense or the exposure time is too long. This irreversible phenomenon is called photobleaching. Such signal loss is a real problem in most imaging experiments, except for some techniques that take advantage of this alteration like FRAP (Fluorescence Recovery After Photobleaching). In the 1970s, FRAP experiments emerged as a new tool for the study of fluorescent protein mobility and dynamics in living cells (Axelrod et al, 1976). Nowadays, it has become a common technique for studying dynamics in almost all aspects of cell biology such as cytoskeleton dynamics (Appaduray et al, 2016), intracellular vesicle transport (Tagawa et al, 2005), cell adhesion (von Wichert, 2003), mitosis (Raccaud et al, 2019) or signal transduction (Giese et al, 2003). Briefly, fluorescent molecules are irreversibly photobleached in regions of interest (ROI) of the cell (cytoplasm, membrane, …) by a high-powered focused laser beam, removing all fluorescent signals (Figure 1A). Subsequent diffusion of surrounding non-bleached fluorescent molecules into the bleached area leads to a recovery of fluorescence (Kang et al, 2010; Lippincott-Schwartz et al, 2018).
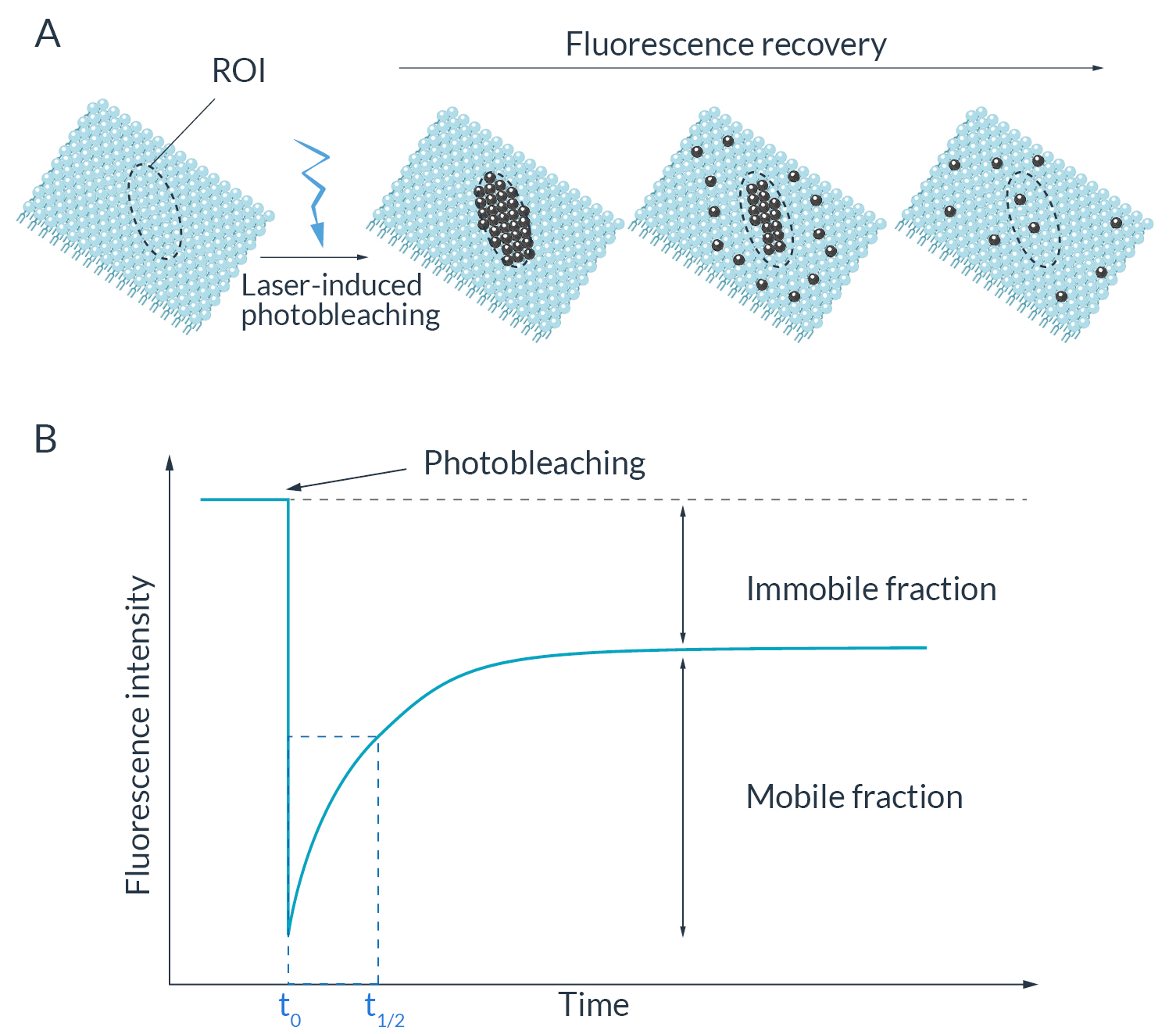
Figure 1. Principle of FRAP imaging
(A) Before the laser-induced photobleaching, the fluorescent proteins are uniformly distributed in a membrane. Immediately after photobleaching, all probes located in the regions of interest (ROI) are not able to emit fluorescence anymore. Return of fluorescence is only due to the exchange of non-fluorescent with fluorescent proteins by lateral diffusion within the membrane. (B) Schematic curve highlighting the fluorescence recovery in time following photobleaching. The immobile fraction is the difference observed between the initial and the final fluorescence intensity. It represents the part of fluorescent protein that is not able to move inside the biological sample observed. The halftime recovery corresponds to the time needed to reach 50% of the final fluorescence intensity (t1/2) following bleaching (t0).
Some parameters can be measured to characterize the dynamics of these fluorescent proteins. One of them is the halftime of recovery (t1/2) which corresponds to the time from the bleach (t0) to the timepoint where the fluorescence intensity reached half of the final recovered intensity (t1/2). The shorter the half-time, the faster the fluorescence recovery occurs and the higher the diffusion. The mobility of fluorescent proteins can also be estimated according to the profile of the intensity recovery curve (Ishikawa-Ankerhold et al, 2012). Mobile and immobile fractions can be measured by calculating the ratios of the final to initial fluorescence intensity (Figure 1B).
To perform FRAP experiments, it is necessary to note that the system has to be adapted to each experiment. For instance, fluorescence recovery could be very fast and needs imaging systems with high frame-rate to improve the temporal resolution of the experiment. Laser control is another key parameter for researchers to adapt, personalize and optimize their ROI selection.
Oxxius lasers for photomanipulation experiments
INSCOPER (Cesson-Sévigné, France) has been working for several years with the laser source designer and manufacturer Oxxius (Lannion, France). This company developed advanced continuous-wave laser modules to target a wide spectrum of applications in biophotonics, spectroscopy, metrology, etc. Oxxius commercializes the L4Cc and L6Cc combiners that are the most compact all-in-one multicolor laser source that could include respectively up to 4 or 6 wavelengths delivered on up to 4 distinct optical fibers outputs. Each laser can be modulated by analog and digital inputs. These controllers can provide a large panel of wavelengths from 375 to 1064nm. Extension modules improve the flexibility of the combiner, by integrating fast switching output ports for FRAP, and adjustable split power for light sheet microscopy among other advanced functionalities.
Inscoper Solution for FRAP imaging
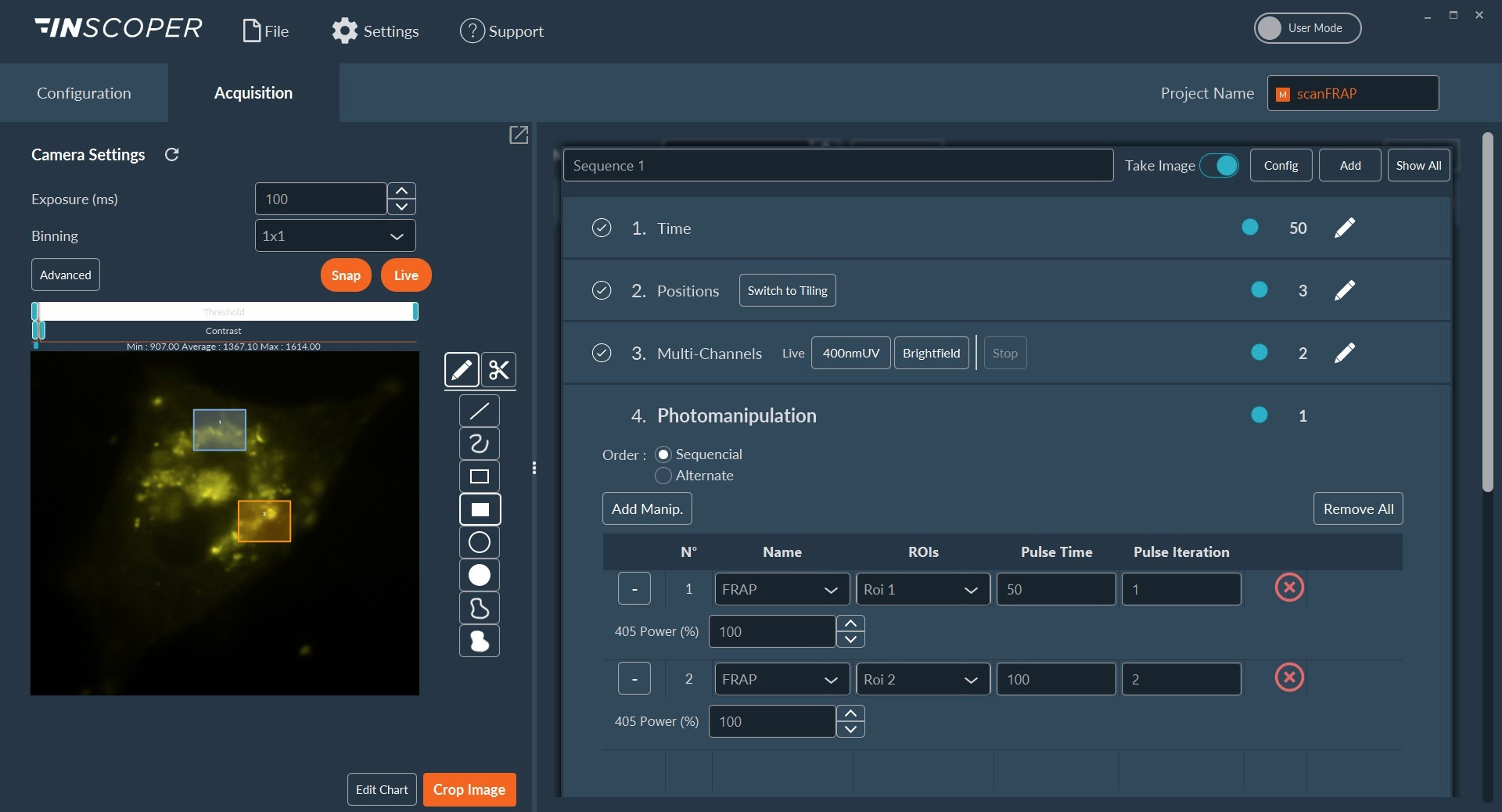
Based on galvanometric mirror technology, the Inscoper scanFRAP is a complete microscopy solution for photomanipulation and optogenetics experiments. The product consists of a software and hardware package compatible with advanced video microscopes used in life science. Incorporating a specially-designed electronic unit to control the microscope stand and third-party devices, the Inscoper scanFRAP provides a new user experience for photomanipulation applications with improved technical performance, full system integration, and ease of use. The core of Inscoper technology eliminates any software latency when controlling the overall microscopy system. It increases the temporal resolution, compared to conventional approaches, which is a major advantage for applications in live cell imaging (Figure 2). Researchers have full control of all laser settings (power, points density, pulse time). They can completely personalize and optimize the bleached areas, modulating the region of interest (ROI) in the software user interface.
Figure 2: Interface of the Inscoper software
Overview of the Inscoper software used to manage the acquisition sequence with multidimensional parameters including timelapse, multiposition, multi-channels, and photomanipulation. All of these dimensions are fully customizable to be more suitable for the user’s experiments.
FRAP experiments
Objectives
Here, we want to characterize the dynamic of the endoplasmic reticulum in living cells using a FRAP approach.
Material
A Nikon Ti2 Eclipse microscope (Nikon, Tokyo, Japan) with a Plan Apo λ 60x 1.4 NA oil immersion objective (MRD01605; Nikon) was used. For this experiment, the camera was a digital CMOS ORCA-Fusion BT (C15440-20UP; Hamamatsu Photonics, Hamamatsu, Japan) and the light engine was from CoolLED (pE-800fura; CoolLED, Andover, United Kingdom). Videomicroscopy and FRAP were performed using Inscoper scanFRAP (INSCOPER, Cesson-Sévigné, France) with a 405nm laser source (L6Cc; Oxxius, Lannion, France). All images were deconvolved using Microvolution software (Microvolution, Cupertino, CA, USA).
Method
HeLa cells were transfected to overexpress YFP (Yellow Fluorescent Protein) in their endoplasmic reticulum. For the FRAP experiment, ROI was defined and bleached using the 405nm laser on living cells and fixed cells as negative control. The fluorescence recovery was then measured. FRAP experiments are basically composed of three steps. First, a short pre-bleached timelapse is captured to get basal levels of fluorescence. Here, a stable and strong YFP signal was observed in transfected cells before photomanipulation. Then, the region of interest (ROI) is selected and photobleached with the appropriate laser settings (wavelength, intensity, point density, …). Finally, another timelapse is performed to monitor in real-time the fluorescence recovery in the bleached ROI.
Results
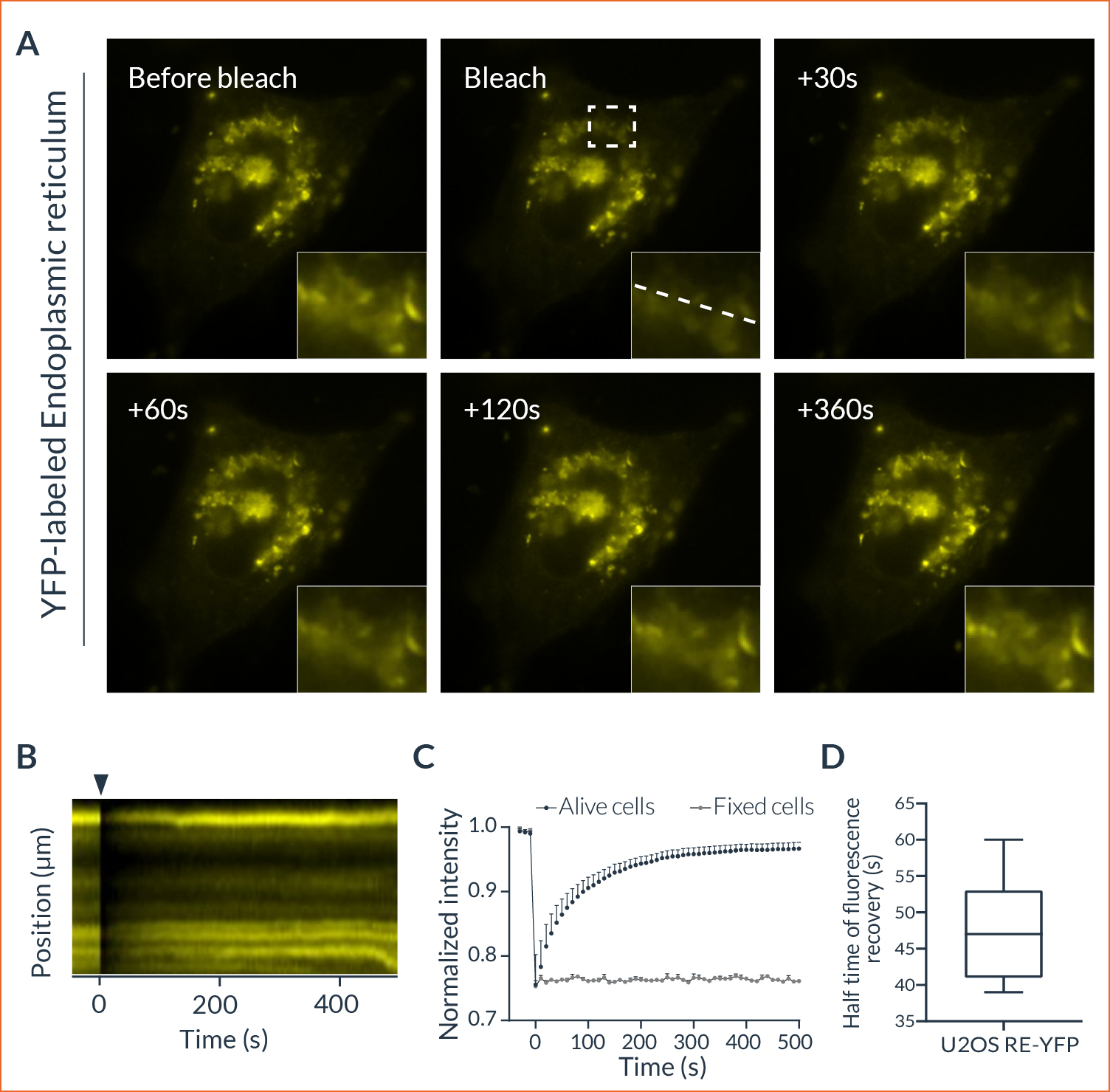
The laser microirradiation represents a powerful tool to monitor DNA repair with high temporal and spatial resolution. DNA lesions were induced following ROI (region of interest) previously defined using the Inscoper software. During the acquisitions, a progressive recruitment of ALC1-GFP protein could be observed over the entire length of the ROI (Figure 3A). Mean intensity of the fluorescent signal has been then measured on the damaged area. The signal from an unaltered ROI was also measured as control. A rapid increase of the signal was observed in the irradiated area, to remain stable in less than 10 seconds (Figure 3B). On the contrary, intensity from the control ROI appeared slightly decreased following the photomanipulation. ALC1-GFP accumulation could also be characterized by the width of the band (Figure 3C). In this experiment, the phenomenon was rapid with a half time of 47.7 ± 7.1 seconds and total recovery in just over 300 seconds (Figure 3D). YFP-labeled proteins from the ER in living cells appeared to be mobile and able to rapidly diffuse inside the cell cytoplasm.
Figure 3: FRAP experiment to monitor the endoplasmic reticulum dynamics in living cells
(A) Representative example of HeLa cells expressing YFP-ER before and after photobleaching. The recovery of the fluorescent signal after photobleaching within the region marked by the dashed rectangle is visualized over time. This area is zoomed in at the bottom right corner of each image. The represented dashed line indicates the area used for the following kymograph. (B) Kymograph representing the evolution of the fluorescence intensity as a function of time. The black arrowhead represents the photobleaching event. (C) Normalized quantification of the fluorescence recovery following a photobleaching event (n = 7). Data are expressed according to the mean ± SEM. (D) Half-time of the fluorescence recovery in FRAP assays (n = 7).
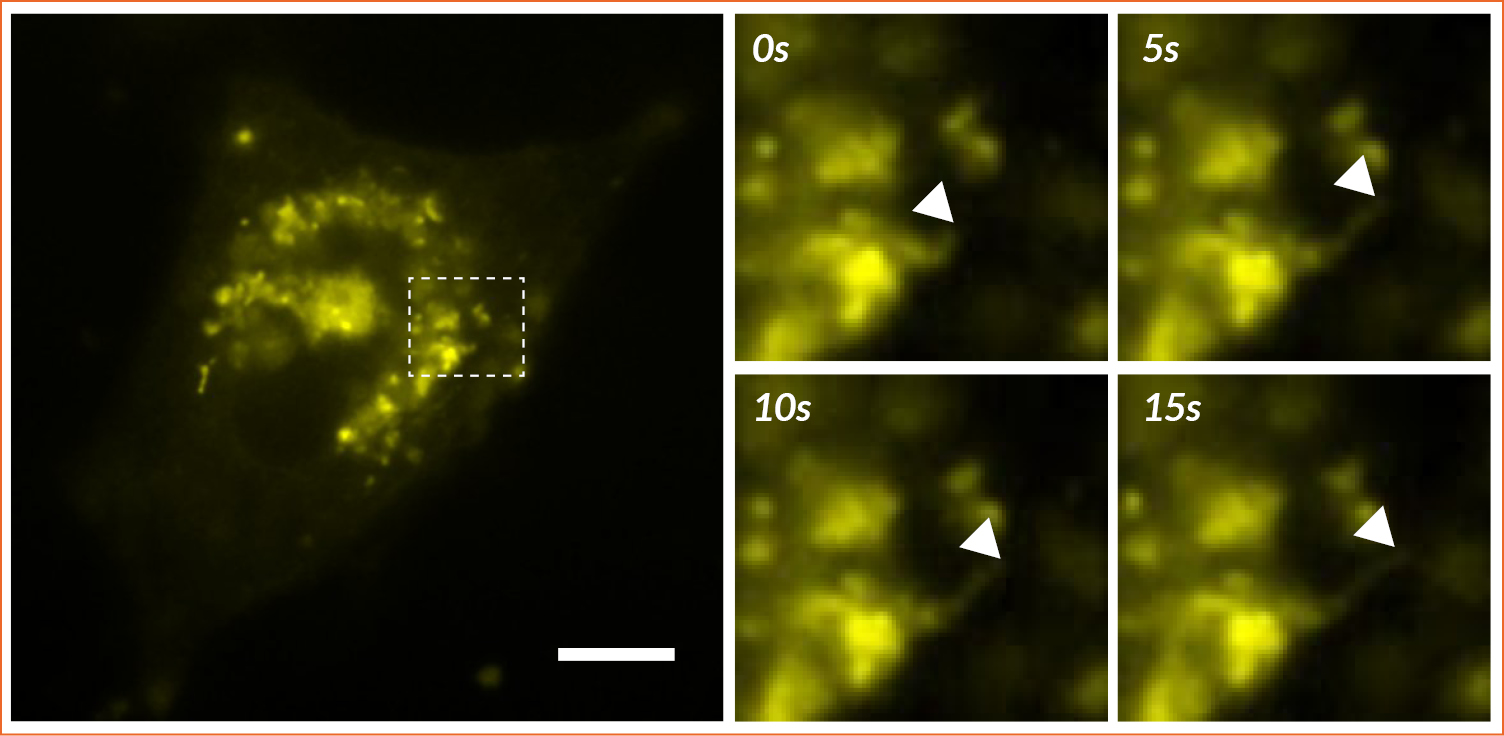
It is important to note that the FRAP approach is not the only technique available to characterize ER dynamics. It can also be monitored by videomicroscopy (Figure 4). Here, ER tubule was imaged, providing users the ability to measure its movement and remodeling. This information is complementary to those previously generated with FRAP, giving information about the formation, fusion/fission of these complex organelles.
Figure 4: Monitoring of endoplasmic reticulum tubule dynamics
Widefield fluorescence imaging of HeLa cells transfected with YFP-ER. The dashed rectangle is zoomed in on the right part of the figure. The elongation of the ER tubule is marked with the arrowhead in the images. Scale bar = 10µm.
Summary
FRAP experiments using the Inscoper scanFRAP offer biologists and microscope users the opportunity to characterize in real-time the kinetics of fluorescence-labeled proteins, combining photomanipulation and videomicroscopy with high spatiotemporal resolution. The scanFRAP stands out from the other products by its customizable features and its smooth integration into a versatile and user-friendly image acquisition software solution. Combined with Oxxius lasers, it is the perfect solution to investigate protein movement/diffusion, compartmentalization, and connections between intracellular compartments, the speed of protein exchange between compartments, and the binding characteristics between proteins. It is worth noting that the Inscoper scanFRAP can be combined with other advanced light imaging techniques (FLIM, FRET), or with various illumination modalities (spinning disk, TIRF, HiLo, light-sheet…) to have a complementary approach.
Bibliography
-
- Appaduray MA, Masedunskas A, Bryce NS, Lucas CA, Warren SC, Timpson P, Stear JH, Gunning PW & Hardeman EC (2016) Recruitment Kinetics of Tropomyosin Tpm3.1 to Actin Filament Bundles in the Cytoskeleton Is Independent of Actin Filament Kinetics. PLOS One 11: 1–16
- Axelrod D, Koppel DE, Schlessinger J, Elson E & Webb WW (1976) Mobility measurement by analysis of fluorescence photobleaching recovery kinetics. Biophys J 16: 1055–1069
- Giese B, Au-Yeung C-K, Herrmann A, Diefenbach S, Haan C, Küster A, Wortmann SB, Roderburg C, Heinrich PC, Behrmann I, et al (2003) Long Term Association of the Cytokine Receptor gp130 and the Janus Kinase Jak1 Revealed by FRAP Analysis. J Biol Chem 278: 39205–39213
- Ishikawa-Ankerhold HC, Ankerhold R & Drummen GPC (2012) Advanced Fluorescence Microscopy Techniques—FRAP, FLIP, FLAP, FRET and FLIM. Molecules 17: 4047–4132
- Kang M, Day CA, DiBenedetto E & Kenworthy AK (2010) A Quantitative Approach to Analyze Binding Diffusion Kinetics by Confocal FRAP. Biophys J 99: 2737–2747
- Lippincott-Schwartz J, Snapp EL & Phair RD (2018) The Development and Enhancement of FRAP as a Key Tool for Investigating Protein Dynamics. Biophys J 115: 1146–1155
- Oakes SA (2020) Endoplasmic Reticulum Stress Signaling in Cancer Cells. Am J Pathol 190: 934–946
- Ozcan L & Tabas I (2012) Role of Endoplasmic Reticulum Stress in Metabolic Disease and Other Disorders. Annu Rev Med 63: 317–328
- Öztürk Z, O’Kane CJ & Pérez-Moreno JJ (2020) Axonal Endoplasmic Reticulum Dynamics and Its Roles in Neurodegeneration. Front Neurosci 14: 1–33
- Raccaud M, Friman ET, Alber AB, Agarwal H, Deluz C, Kuhn T, Gebhardt JCM & Suter DM (2019) Mitotic chromosome binding predicts transcription factor properties in interphase. Nat Commun 10: 1–16
- Roussel BD, Kruppa AJ, Miranda E, Crowther DC, Lomas DA & Marciniak SJ (2013) Endoplasmic reticulum dysfunction in neurological disease. Lancet Neurol 12: 105–118
- Tagawa A, Mezzacasa A, Hayer A, Longatti A, Pelkmans L & Helenius A (2005) Assembly and trafficking of caveolar domains in the cell. J Cell Biol 170: 769–779
- von Wichert G (2003) Force-dependent integrin-cytoskeleton linkage formation requires downregulation of focal complex dynamics by Shp2. EMBO J 22: 5023–5035

0 Comments